The long delay for the publication of this and the previous blog post is due to my time-consuming work for the following piece for my online Honors Chemistry class. Studying agrochemicals is not exactly what one might call a delightful or uplifting study, but I do believe that it’s immensely important. I would encourage you to independently research the practices of the commercial farms providing you and your children with food. When you come to a point of utter discouragement and your stomach begins to tighten, stop by Joel Salatin’s Polyface Farm and be sure to view their movie clips. What a refreshing breath of air! I am encouraged to continue doing my best with the backyard vegetable plot.
The purpose of this paper is to examine two commonly used agrochemicals. The commercial farm has been under attack by various thinkers concerned with the extensive use of potentially dangerous chemicals in the production of food for human consumption. What are these chemicals, what role do they play, and how do they actually affect the health of humans and the environment? Finally - what are we doing about it?
Ammonia
What is ammonia?
Ammonia is a colorless, caustic gas with a biting, suffocating odor. It is produced naturally by the decomposition of organic matter (plants, animals, feces) and plays an essential role in biology and the nitrogen cycle. Ammonia is also one of the most widely manufactured inorganic chemicals. It is processed by the Haber-Bosch method, a simple synthesis reaction written N2(g) + 3H2(g) → 2NH3(g). This simply means that one mole of nitrogen gas reacts (with the aid of an iron catalyst) with three moles of hydrogen gas to produce two moles of ammonia gas.
The molecular model for this reaction is pictured below:

How is ammonia used in agriculture?
The Haber-Bosch process is vital to the agricultural industry as one of the most readily available mass-producers of ammonia. Of the 100 million tons of ammonia commercially produced worldwide each year, eighty percent is used as fertilizer. Pure ammonia, ammonia hydroxide, or fertilizers with ammonium compounds are applied to fields to increase the nitrogen levels in the soil. Anhydrous ammonia is also used to control mold growth in high-moisture grains, corn silage, and hay fed to livestock.
The commercial farm is also a significant center of natural ammonia production, since the decomposition of animal manure releases this gaseous chemical. The feeding and housing methods characteristic of commercial farms actually elevate these ammonia emissions to unnatural levels, as will be explored in a later section.
The Larger Good?
Ammonia is immensely valuable to the commercial farmer, who is often cultivating vast fields of a single crop. Rich in nitrogen, it reduces the need for crop rotation, allowing the farmer not only to utilize as much land as possible, but to develop his own niche in the highly competitive agricultural industry.
Ammonia and other fertilizers are added to the soil to increase food production and quality (i.e., uniformity and good looks). As the world is becoming increasingly industrialized and the countryside is taken up by development, farmers are faced with the dilemma of fewer people producing more food on less land. Commercial farmers have largely abandoned the wise, traditional practices of crop rotation and diversity for the more expedient modern method of rigorous chemical fertilization. After many years of such poor treatment, however, the soil is left worn and depleted.
Environmental and Health Concerns
Ammonia is a natural component of a healthy environment and biology, but increasing levels of ammonia in the atmosphere and water are causing problems, In fact, the chemical has been classified as “dangerous to the environment. Ammonia has been identified as a major global air quality concern by the National Research Council. Volatilized ammonia emissions are able to travel hundreds of miles from their origin, leading to the pollution of even distant environments. Ammonia can also react with other compounds to form particulate matter with a diameter of up to 2.5 microns. This tiny diameter allows them to penetrate deep into the lungs, causing significant respiratory and cardiovascular problems. Long-term exposure to PM2.5 may lead to premature death.
Ammonia has been responsible for the eutrophication (excessive nutrient enrichment) of surface waters, which promotes harmful algae growth and kills aquatic animals. Even dilute concentrations of ammonia are highly toxic to fish and amphibians, which have none of the biological defenses as have mammals. Many have linked the global decrease in the frog population to excessive ammonia.
Excessive ammonia exposure also poses a direct danger to human health. It is an extremely caustic chemical and anhydrous ammonia may cause severe dehydration and chemical burns when contacting the skin. Even short exposure to low levels of ammonia gas create health problems, particularly lung and eye irritation. In extreme cases, blindness, permanent lung damage, and even death can occur.

Despite these disreputable practices, much of the environmental concerns regarding ammonia are directed, ironically enough, to the natural production of ammonia in animal wastes. Even so, such attention is not wholly misdirected.
The content of ammonia in manure has been significantly increased by the practice of feeding animals high protein feed containing a surfeit of nitrogen. (This is the pleasanter way to describe a feedlot diet of chicken feathers, dung, rendered carcasses, and blood.) The animals’ inability to efficiently convert the high levels of nitrogen into animal products results in nitrogen-rich excretion. As the manure decomposes, microbial action releases ammonia into the air. Commercial farm animals are often continuously housed in closed quarters with hundred of other animals. Manure build-up creates high levels of ammonia gases. This is dangerous to the health of the workers, the animals, and those who eat the products of those animals.
Solutions and Precautions
In the production and handling of ammonia, safety equipment is carefully regulated. Workers must wear protective clothing, and all containers and systems must meet safety standards. The U.S. Occupational Safety and Health Administration (OSHA) has established an exposure limit of 15 minutes for gaseous ammonia of 35 ppm. A hazardous safety permit is required for transporting large amounts of ammonia.
There is no prescribed cure for ammonia poisoning. Fortunately, the human body is equipped with defenses that aid in the excretion of ammonia or in its transformation to harmless substances. Doctors treat the symptoms of ammonia poison and the victim usually recovers on his own.
Notwithstanding the alarming facts, the Institute for Food and Agricultural Science still advocates the use of ammoniated fodder with non-lactating livestock. This is indeed an improvement, but the evidence indicates that the treated food is damaging to the general health of livestock in all stages of life.
Again, much concern and research is dedicated to the natural ammonia emissions of livestock. Researchers have submitted two principle ways to reduce ammonia emissions from manure. The first involves adding acid-producing phosphorus sources, calcium chloride, or calcium sulfate to animal feed. Another popular method involves a post-excrement procedure in which aluminum sulfate, ferrous sulfate, phosphoric acid, or calcium salts are added to the manure where the animals are housed. Authorities have also cited the need to install sufficient vents for proper air circulation.
Yet these precautions largely address the symptoms and not the cause. The only long-term remedy would seem to be a return to traditional farming practices, a radical step from the modern industry model. Cows, for example, should be allowed to forage freely in grass pastures rather that be tied in crowded, poorly ventilated feedlots, eating food nature never intended them to eat. The ammonia content in their manure would return to normal levels and could then be used to dress the fields. Granted, this natural fertilizer is not strong enough to support the present mass-production. We would have to return to the more natural practices of crop rotation and crop diversity.
This system, of course, would hardly be practical for the mainstream agribusiness competing in an aggressive industry. This is, in part, my contention. People are much too willing to sacrifice health and quality to economic expediency and convenience. Until concerned individuals use their voices, pens, and pocket-books to combat these practices, things will remain much the same.
Remember...
I wish to stress again that ammonia plays a natural and vital role in biological and environmental health. The problem comes when we disrupt the natural balance by the widespread production and use of this chemical.
Organophosphates
What are organophosphates?
Organophosphates are synthetic chemicals; that is, they are not produced in nature. They are widely used because the organic groups can be so easily linked. They are also acute neuro-toxins once used in chemical warfare. Fortunately, organophosphates break down rapidly.
“Organophosphates” is actually a general term for esters of phosphoric acid. There are several variations of the chemical, as can be represented by the three following equations:
OP(OH)3 + ROH → OP(OH)2(OR) + H2O
OP(OH)2(OR) + R'OH → OP(OH)(OR)(OR') + H2O
OP(OH)(OR)(OR') + R"OH → OP(OR)(OR')(OR") + H2O
In words, phosphoric acid reacts with an alcohol to produce organophosphates and water.
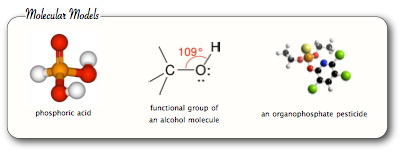
How are organophosphates used in agriculture?
Organophosphorus compounds are widely used as pesticides both in agrarian and residential settings. They comprised 80% of worldwide insecticide sales, and are also widely used as sheep and cattle dips to prevent such external parasites as ticks, ich mites, blow-flies, keds, lice, and warble flies.
Organophosphates work by irreversibly disabling acetylcholinesterase, an enzyme essential to nerve function. This not only works in the intended insect victims, but in other animals and in humans.
The Larger Good?
Organophosphates and other insecticides are used extensively by farmers to prevent crop losses due to harmful insects. Organophosphates are also used in livestock dips to deter problems caused by external parasites.
Environmental and Health Concerns
The use of insecticides, particularly such acute neurotoxins as organophosphorus, is linked to lasting neurological problems for farmers, including recurring headaches, dizziness, nausea, fatigue, insomnia, hand tremors, and numbness. Because organophosphates are readily absorbed through the skin, lungs, and gastrointestinal tract, they are especially dangerous. Chronic exposure to organophosphate can cause confusion, anxiety, loss of memory, loss of appetite, disorientation, depression, and personality changes. Exposure to organophosphates has also been linked to reproductive problems: infertility, miscarriage, stillbirth, pre-term delivery, low birth weight, and birth defects. The chances of miscarriage are increased by up to 129%.

In order to eliminate pests, animals are completely immersed in a toxic brew of pesticides and fungicides. Organophosphates are absorbed into the body, and studies have shown that OP residues may contaminate cows’ milk after treatment. Many of these compounds can also cross the placental membranes, causing toxicity in offspring. Mark Purdy has famously linked organophosphates to Mad Cow Disease. Sometimes such direct exposure is not the only danger. Research studies have linked Mad Cow Disease to the practice of using treated fruit peel as cattle fodder.
Chemical pesticides are a particular threat against animal health and life. Organophosphates in particular can become toxically addictive to wildlife. Insecticides have been shown to kill bees and perhaps other pollinators, which will mean a reduction in crop yields. The USDA and USWS estimate that U.S. farmers loose at least $200 yearly through pollinator decline due to the use of pesticides. Not only are we destroying beneficial insects, but we are also breeding OP-resistant insects, which will call for even stronger insecticides and make it more and more difficult to return to natural farming.
The short-term benefit of fewer insect pests will lead to ultimate problems of soil deficiency and pollinator decline, not to mention long-term delayed neuro-toxic damage to both animals and humans. Is the momentary convenience really worth it? Who will pay for the damages?
Solutions and Precautions
Farmers and others working with pesticides are required to use protective clothing and respiratory protection, but studies have shown that even licensed applicators are betraying neurological symptoms.
Concerned individuals are turning to organic diets to reduce their exposure to pesticides. Studies have shown that those who eat organic diets are far less exposed to organophosphates in their food than those who eat the conventional counterparts.
Fortunately, some concerned individuals and organizations are working to further public education on the possibility of OP and pesticide alternatives. Researchers, for example, are exploring the use of pheromones to impede insect mating, though such an approach has proven controversial. The introduction of natural predators is also an option, though care must be taken not to disrupt the environment’s food web. Crop rotation and diversity are natural, traditional farming practices that also help to prevent any real insect epidemics.
In Conclusion
The world of chemistry offers exciting new frontiers and valuable possibilities, but caution and thought must be exercised as we advance. Oftentimes the excitement of immediate benefit blinds us to the ultimate consequences. People today are concerned with being able to feed the global population. As has been demonstrated, although the use of chemical fertilizers and pesticides, as well as growth hormones and herbicides, may bring immediate increase, it may very well lead to major deficiencies and problems. We need to weigh our choices more carefully, not in light of monetary profit or momentary convenience, but in light of the coming generations who will inherit the earth we help to form.
Image Credits
Title Photographs: Tomatoes (www.thisoldhouse.com); Jersey Heifer (www.womeninthebible.net); Grain (www.iwxm.net); Watering (www.wikipedia.org). Molecular Models: Nitrogen, Ammonia, Phosphoric Acid, Alcohol (www.wikipedia.org); Hydrogen (www.princeton.edu); Organophosphate Pesticide (http://www.panna.org). Illustrations: Ammoniating Hay (http://pubs.caes.uga.edu/caespubs/pubcd/L402.htm); Sheep Dip (http://www.mfe.govt.nz/).